|
Scan the films at high enough DPI t avoid pixelated bands, or capture a high resolution exposure for IR imaging methods. It greatly improves consistency.ĭevelop your blots with similar times according to your dilution curve or empirical experience, as above. This normalizes the current for membrane area and transfer time. Load equal sample masses and transfer with constant current, using same mW*hr/cm 2 of membrane to deposit similar amounts of protein onto your membranes. It starts with the PAGE/transfer process. Here's how I do it when I need to quantify bands for biological replicates that are run on different membranes. It's not even worth quantifying saturated bands. Then establish/optimize the antibody concentrations, wash times/buffers, and development time.Īt a bare minimum make sure that you haven't saturated your signal (a max/min pixel value for your LUTs on imageJ, for example). So a good example for a mini gel would be 25ug lysate, 12.5ug, 6.25, 3.125, etc. This can be as simple as loading a serial dilution series of your lysate and then identifying an exposure range for film or IR detection that gives you measurable, non-saturated intensities for dilutions you usually load. Preface: It's best to find your linear range before even attempting quantification. Quantification is always the way to go, so you're on the right path. You want to be able to say that treatment led to a 2 or 3-fold change in fluroescence when you write it up, rather than just give a qualitative statement. You may not have to actually show it in the final figure, but just have it for yourself or for reviewers. I cannot recommend it enough if you have access, and if you do I would be happy to tell you all about it.Īs for whether or not this analysis is necessary, I would say that it is. 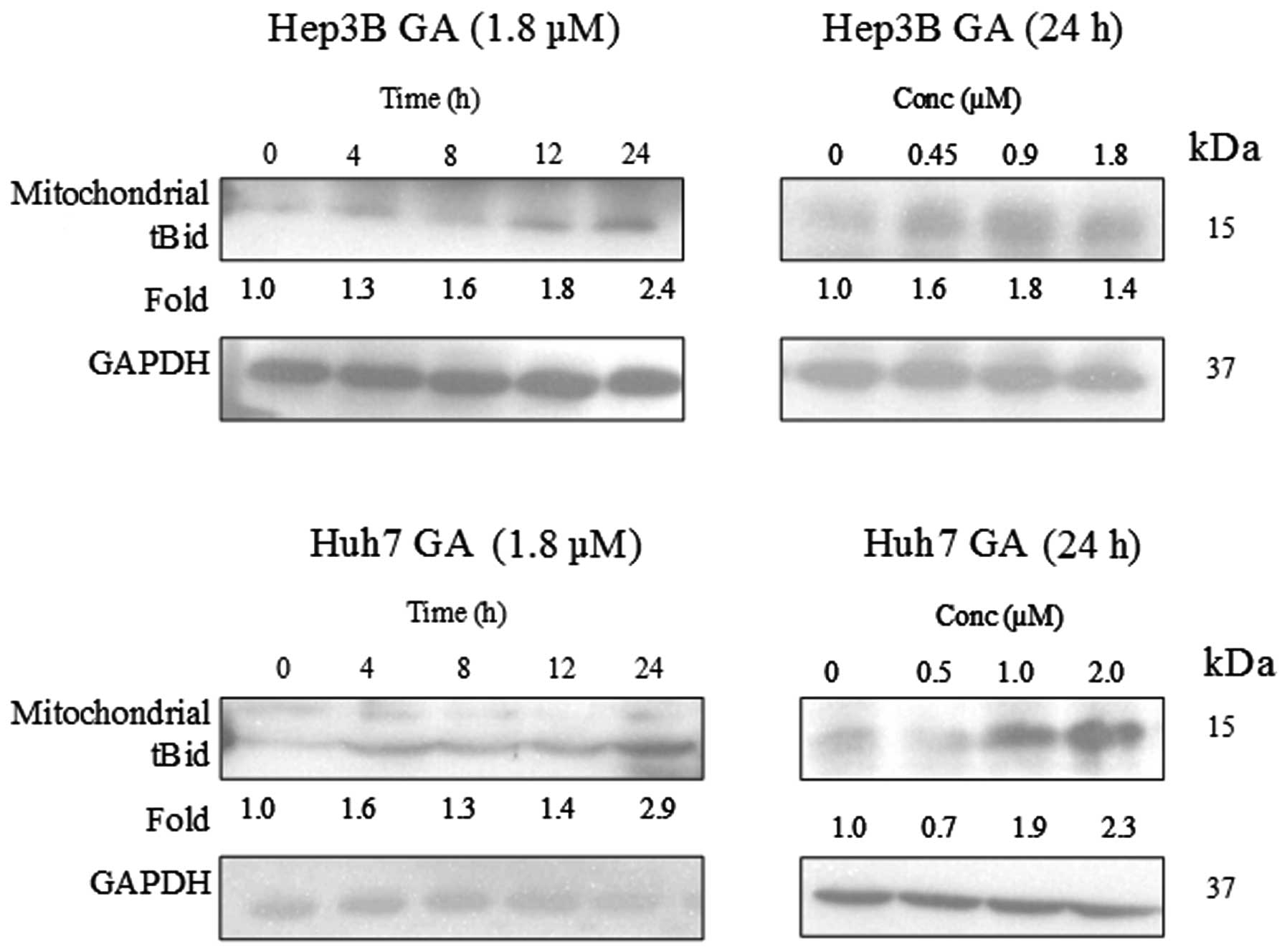
Whenever I want to quantify protein that is at least somewhat abundant, I use fluorescent western blots with the LICOR Odyssey system. This is the guide I used in the past for ImageJ and chemiluminescent blots This is a reason I moved away from chemiluminescence to fluorescence, especially for for quantification.Ģ - Im wondering if you did something different in the analysis. Two things:ġ - The more intense bands look saturated, you should try a shorter exposure for quantification. The difference between those bands looks SO much bigger than D7. First thing I noticed - Unless I'm reading this incorrectly, there is NO way D3 is only a 2-fold change (or maybe its closer to 3-fold, but its hard to tell with how you've presented the data).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
